Peripheral T-Cell Lymphoma Market to Witness Significant Expansion at a CAGR of 7% During the Forecast Period (2026–2036) Amid Advancing Treatment Landscape | DelveInsight

The peripheral T-cell lymphoma treatment market is witnessing steady growth, primarily driven by the increasing incidence, leading to an increased patient population, which fuels the ongoing treatment demand. Additionally, the launch of emerging therapies such as Duvelisib (COPIKTRA) (Secura Bio), Lacutamab (IPH4102) (Innate Pharma), Linperlisib (YY-20394/linprixetine) (Shanghai YingLi Pharmaceutical), KEYTRUDA (Merck), OPDIVO (BMS), Golidocitinib (AZD4205/DZD4205) (Dizal Pharmaceuticals), and others will further propel the market growth forward in the coming years.
New York, USA, April 27, 2026 (GLOBE NEWSWIRE) — Peripheral T-Cell Lymphoma Market to Witness Significant Expansion at a CAGR of 7% During the Forecast Period (2026–2036) Amid Advancing Treatment Landscape | DelveInsight
The peripheral T-cell lymphoma treatment market is witnessing steady growth, primarily driven by the increasing incidence, leading to an increased patient population, which fuels the ongoing treatment demand. Additionally, the launch of emerging therapies such as Duvelisib (COPIKTRA) (Secura Bio), Lacutamab (IPH4102) (Innate Pharma), Linperlisib (YY-20394/linprixetine) (Shanghai YingLi Pharmaceutical), KEYTRUDA (Merck), OPDIVO (BMS), Golidocitinib (AZD4205/DZD4205) (Dizal Pharmaceuticals), and others will further propel the market growth forward in the coming years.
Recently published PTCL Market Insights report includes a comprehensive understanding of current treatment practices, peripheral T-cell lymphoma emerging drugs, market share of individual therapies, and current and forecasted market size from 2022 to 2036, segmented into leading markets [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
Peripheral T-Cell Lymphoma Market Summary
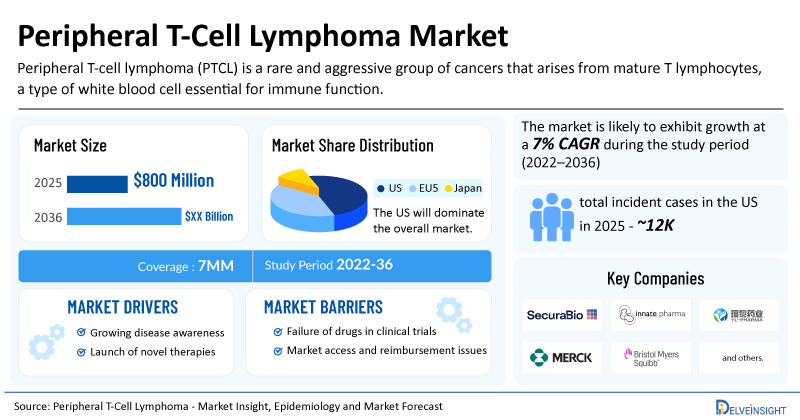
- The market size for peripheral T-cell lymphoma was found to be USD 800 million in the 7MM in 2025.
- The United States accounted for the largest peripheral T-cell lymphoma treatment market size in 2025, i.e 68%, compared to other major markets, including the EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- In the 7MM, ADCETRIS holds the largest market share in 2025, generating over USD 300 million.
- The US accounted for about 12,000 incident cases of PTCL in 2025.
- Key peripheral T-cell lymphoma companies, including Secura Bio, Innate Pharma, Shanghai YingLi Pharmaceutical, Merck, BMS, Dizal Pharmaceuticals, and others, are actively working on innovative peripheral T-cell lymphoma drugs.
- Some of the key peripheral T-cell lymphoma therapies in clinical trials include Duvelisib (COPIKTRA), Lacutamab (IPH4102), Linperlisib (YY-20394/linprixetine), KEYTRUDA, OPDIVO, Golidocitinib (AZD4205/DZD4205), and others. These novel peripheral T-cell lymphoma therapies are anticipated to enter the peripheral T-cell lymphoma market in the forecast period and are expected to change the market.
Discover which region dominates the PTCL market @ https://www.delveinsight.com/sample-request/peripheral-t-cell-lymphoma-ptcl-market
Key Factors Driving the Growth of the Peripheral T-Cell Lymphoma Market
- Rising PTCL Incidence Fueling Market Growth: The increasing incidence of PTCL across the 7MM is driven by a combination of apparent epidemiologic inflation and true biological change, rather than a single causative factor. One of the most important contributors is improved disease recognition and classification. In the US, in 2025, there were ~12,000 incident cases of PTCL, which will increase by 2036.
- Rising Opportunities in R/R Patient Pool Contributing to Increase in PTCL Market: The recent introduction of therapies such as HIYASTA for ATLL and PTCL, along with DARVIAS and REMITORO for R/R ATLL and PTCL in Japan, has expanded the treatment landscape and provided additional therapeutic options for patients with PTCL.
- Advancements in CD30-Directed Treatment Strategies Boosting the PTCL Market: Novel therapies targeting CD30 include chimeric antigen receptor therapies and bispecific antibody T-cell engagers, which may be expected to further improve outcomes for patients with CD30-positive lymphomas in the coming years.
- Advancing Therapeutics for EBV-Driven T-Cell Lymphomas: Emerging therapies such as Viracta Therapeutics’ Nanatinostat are the only ones being developed for EBV+ T cell lymphomas in a possible Phase II registrational trial.
- Launch of Emerging PTCL Drugs Expected to Increase Market: The dynamics of the PTCL market are expected to change in the coming years due to the launch of emerging therapies such as Duvelisib (COPIKTRA) (Secura Bio), Lacutamab (IPH4102) (Innate Pharma), Linperlisib (YY-20394/linprixetine) (Shanghai YingLi Pharmaceutical), KEYTRUDA (Merck), OPDIVO (BMS), Golidocitinib (AZD4205/DZD4205) (Dizal Pharmaceuticals), and others.
Sadaf Javed, oncology expert at DelveInsight, said that with multiple late-stage and mid-stage assets progressing amid a small, fragmented market, future success will favor therapies that demonstrate clear superiority over existing agents, biomarker-aligned positioning, and scalable commercialization strategies from financially resilient players.
Peripheral T-Cell Lymphoma Market Analysis
- Treatment approaches for peripheral T-cell lymphoma (PTCL) primarily involve combination chemotherapy regimens such as CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) and CHOEP (which adds etoposide), along with localized radiotherapy, stem cell transplantation, and corticosteroid-based therapies.
- In terms of approved therapies, the U.S. FDA has authorized drugs including BELEODAQ (belinostat), FOLOTYN (pralatrexate), ARRANON (nelarabine), XALKORI (crizotinib), and ADCETRIS (brentuximab vedotin) for PTCL management.
- In Europe, ADCETRIS and ARRANON are approved, while Japan has a broader range of options such as ISTODAX (romidepsin), FOLOTYN (pralatrexate), POTELIGEO (mogamulizumab), ADCETRIS (brentuximab vedotin), MUNDESINE (forodesine hydrochloride), ARRANON (nelarabine), HIYASTA (tucidinostat), DARVIAS (darinaparsin), EZHARMIA (valemetostat tosylate), and REMITORO (denileukin diftitox).
- The pipeline for PTCL is also evolving, with promising investigational therapies such as Linperlisib (Shanghai YingLi Pharmaceutical), Golidocitinib (Dizal Pharmaceutical), Lacutamab (Innate Pharma), and AUTO4 (Autolus Therapeutics). Additional candidates under development across the 7MM include GFH009 (GenFleet Therapeutics) and PTX-100 (Prescient Therapeutics), among others.
- Tucidinostat, originally developed by Chipscreen, was licensed to HUYABIO for ex-China markets in 2007. It received approval in China as Chidamide (EPIDAZA) in 2014 and later in Japan as HIYASTA in 2021 for relapsed or refractory PTCL.
- Overall, the PTCL treatment landscape is expected to undergo substantial transformation between 2026 and 2036. Advances in targeted therapies and biomarker-driven approaches are likely to improve patient outcomes, both as standalone treatments and in combination regimens, offering a more optimistic outlook for individuals with PTCL.
Learn more about the upcoming approvals in the PTCL market @ Peripheral T-Cell Lymphoma Treatment Market
Peripheral T-Cell Lymphoma Competitive Landscape
Some of the PTCL drugs under development include Duvelisib (COPIKTRA) (Secura Bio), Lacutamab (IPH4102) (Innate Pharma), Linperlisib (YY-20394/linprixetine) (Shanghai YingLi Pharmaceutical), KEYTRUDA (Merck), OPDIVO (BMS), Golidocitinib (AZD4205/DZD4205) (Dizal Pharmaceuticals), and others.
Secura Bio’s Duvelisib (COPIKTRA) is an orally administered inhibitor of phosphoinositide 3-kinase (PI3K). Activation of the PI3K pathway is associated with the growth of malignant cells and is believed to contribute to the development and persistence of a supportive tumor microenvironment. In December 2025, Secura Bio reported that its Phase III TERZO trial is progressing as planned, with an interim data readout expected in early 2027 based on current enrollment; the study was initiated earlier in the year, with the first patient dosed in June.
Innate Pharma’s Lacutamab is a first-in-class, humanized antibody targeting KIR3DL2 that induces cytotoxicity. It is currently under evaluation in an open-label, multicohort Phase II study in CTCL, as well as a separate Phase II trial in PTCL. In December 2025, Innate Pharma indicated that data from the Phase II PTCL study are expected by the end of 2026.
Shanghai YingLi Pharmaceutical’s Linperlisib is a highly selective and potent PI3Kδ inhibitor with a favorable safety profile. It has demonstrated encouraging anti-tumor activity along with promising pharmacokinetic and pharmaceutical characteristics as a once-daily oral therapy in clinical development. The drug has been evaluated across three clinical trials in relapsed/refractory PTCL in China, the United States, and Europe, involving more than 165 treated patients.
Dizal Pharmaceuticals’ Golidocitinib is currently the first and only selective Janus kinase 1 (JAK1) inhibitor under investigation for the treatment of relapsed/refractory PTCL. Phase I results (JACKPOT8 Part A) have been published in Annals of Oncology, while data from the global pivotal trial (JACKPOT Part B) were published in The Lancet Oncology.
The anticipated launch of these emerging peripheral T-cell lymphoma therapies are poised to transform the peripheral T-cell lymphoma market landscape in the coming years. As these cutting-edge peripheral T-cell lymphoma therapies continue to mature and gain regulatory approval, they are expected to reshape the peripheral T-cell lymphoma market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about top companies developing PTCL drugs, visit @ Peripheral T-Cell Lymphoma Medication
Recent Developments in the Peripheral T-Cell Lymphoma Market
- In March 2026, Corvus announced that it continues to enroll patients in a registrational Phase III clinical trial of soquelitinib in patients with R/R PTCL at multiple clinical sites. The FDA has granted soquelitinib Orphan Drug Designation (ODD) for the treatment of T cell lymphoma and Fast Track designation (FTD) for the treatment of adult patients with relapsed or refractory PTCL after at least two lines of systemic therapy.
- In December 2025, Secura Bio presented extended follow-up analyses examining additional safety data and the impact of prior lines of therapy from the company’s Phase II PRIMO trial (NCT03372057) at the 2025 American Society of Hematology (ASH) Annual Meeting. The results continue to show that duvelisib delivers meaningful clinical activity with a manageable safety profile, even in heavily pretreated patients, underscoring its potential role across a broad PTCL population.
- In December 2025, Dizal Pharmaceutical presented new data of golidocitinib at the 67th ASH annual meeting with results from newly diagnosed PTCL, R/R PTCL, rare subtypes of PTCL, and PTCL-associated HLH.
What is Peripheral T-Cell Lymphoma?
Peripheral T-cell lymphoma (PTCL) is a rare and aggressive group of cancers that arises from mature T lymphocytes, a type of white blood cell essential for immune function. It falls under the broader category of non-Hodgkin lymphoma and encompasses several biologically diverse subtypes, such as PTCL-not otherwise specified (PTCL-NOS), angioimmunoblastic T-cell lymphoma (AITL), and anaplastic large cell lymphoma (ALCL). PTCL typically presents with symptoms like enlarged lymph nodes, fever, weight loss, and night sweats, often indicating advanced disease at diagnosis. Compared to B-cell lymphomas, PTCL generally has a poorer prognosis and limited treatment options, with standard therapies including combination chemotherapy, targeted agents, and, in some cases, stem cell transplantation.
Peripheral T-Cell Lymphoma Epidemiology Segmentation
The peripheral T-cell lymphoma patient pool analysis section provides insights into the historical and current peripheral T-cell lymphoma patient pool and forecasted trends for the leading markets. In Japan, the increase in PTCL incidence is driven by the Endemic HTLV-1 infection, particularly in southwestern Japan, which directly increases the incidence of adult T-cell leukemia/lymphoma (ATL), a major PTCL subtype, contributing to higher overall PTCL rates.
The peripheral T-cell lymphoma market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Total Incident Cases of PTCL
- Stage-specific Incident Cases PTCL
- Type-specific Incident Cases of PTCL
- CD30 Expression-specific Incident Cases PTCL
- Line-wise Treated Cases of PTCL
Download the report to understand unmet needs in PTCL therapy @ Peripheral T-Cell Lymphoma Treatment Options
| Peripheral T-Cell Lymphoma Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Peripheral T-Cell Lymphoma Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Peripheral T-Cell Lymphoma Market CAGR | 7% |
| Peripheral T-Cell Lymphoma Market Size in 2025 | USD 800 Million |
| Key Peripheral T-Cell Lymphoma Companies | Secura Bio, Innate Pharma, Shanghai YingLi Pharmaceutical, Merck, BMS, Dizal Pharmaceuticals, Daiichi Sankyo, Pfizer, HUYA Bioscience International (HUYABIO), and others |
| Key Peripheral T-Cell Lymphoma Therapies | Duvelisib (COPIKTRA), Lacutamab (IPH4102), Linperlisib (YY-20394/linprixetine), KEYTRUDA, OPDIVO, Golidocitinib (AZD4205/DZD4205), EZHARMIA, XALKORI, HIYASTA, and others |
Scope of the Peripheral T-Cell Lymphoma Market Report
- Peripheral T-Cell Lymphoma Therapeutic Assessment: Peripheral T-Cell Lymphoma current marketed and emerging therapies
- Peripheral T-Cell Lymphoma Market Dynamics: Conjoint Analysis of Emerging Peripheral T-Cell Lymphoma Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Peripheral T-Cell Lymphoma Market Unmet Needs, KOL’s views, Analyst’s views, Peripheral T-Cell Lymphoma Market Access and Reimbursement
Discover more about PTCL pipeline drugs 2026 @ Peripheral T-Cell Lymphoma Clinical Trials
Table of Contents
| 1 | Peripheral T-Cell Lymphoma Market Key Insights |
| 2 | Peripheral T-Cell Lymphoma Market Report Introduction |
| 3 | Executive Summary |
| 4 | Key Events |
| 5 | Epidemiology and Market Forecast Methodology |
| 6 | Peripheral T-cell lymphoma (PTCL) Market Overview at a Glance |
| 7 | Disease Background and Overview of Peripheral T-cell Lymphoma (PTCL) |
| 7.1 | Introduction |
| 7.2 | Causes and Risk Factors |
| 7.3 | Signs and Symptoms |
| 7.4 | Pathophysiology |
| 7.5 | Diagnosis of Peripheral T-cell lymphoma (PTCL) |
| 8 | Epidemiology and Patient Population of Peripheral T-cell lymphoma (PTCL) |
| 8.1 | Key Findings |
| 8.2 | Assumptions and Rationale |
| 8.3 | Total Incident Cases of Peripheral T-cell lymphoma (PTCL) in the 7MM |
| 8.4 | The United States |
| 8.4.1 | Total Incident cases of Peripheral T-cell lymphoma (PTCL) in the US |
| 8.4.2 | Stage-specific Incident Cases of Peripheral T-cell lymphoma (PTCL) in the US |
| 8.4.3 | Type-specific Incident Cases of Peripheral T-cell lymphoma (PTCL) in the US |
| 8.4.4 | CD30 Expression-specific Incident Cases of Peripheral T-cell lymphoma (PTCL) in the US |
| 8.4.5 | Line-wise Treated Cases of Peripheral T-cell lymphoma (PTCL) in the US |
| 8.5 | EU4 and the UK |
| 8.6 | Japan |
| 9 | Patient Journey of Peripheral T-cell lymphoma (PTCL) |
| 10 | Marketed PTCL Therapies |
| 10.1 | Marketed Competitive Landscape of PTCL |
| 10.2 | Valemetostat tosilate (EZHARMIA): Daiichi Sankyo |
| 10.2.1 | Product Description |
| 10.2.2 | Regulatory Milestones |
| 10.2.3 | Other Developmental Activities |
| 10.2.4 | Summary of Pivotal Trials |
| 10.2.5 | Analyst’s View |
| 10.3 | Crizotinib (XALKORI): Pfizer |
| 10.4 | Tucidinostat (HIYASTA): HUYA Bioscience International (HUYABIO) |
| List to be continued in the report… | |
| 11 | Emerging PTCL Therapies |
| 11.1 | Emerging Competitive Landscape of PTCL |
| 11.2 | Duvelisib (COPIKTRA): Secura Bio |
| 11.2.1 | Product Description |
| 11.2.2 | Other Developmental Activities |
| 11.2.3 | Clinical Development |
| 11.2.3.1 | Clinical Trial Information |
| 11.2.4 | Safety and Efficacy |
| 11.2.5 | Analyst Views |
| 11.3 | Lacutamab (IPH4102): Innate Pharma |
| 11.4 | Linperlisib (YY-20394/linprixetine): Shanghai YingLi Pharmaceutical |
| 11.5 | KEYTRUDA: Merck |
| 11.6 | OPDIVO: BMS |
| 12 | Peripheral T-cell lymphoma (PTCL) Market: 7MM Analysis |
| 12.1 | Key Findings |
| 12.2 | Market Outlook of Peripheral T-cell lymphoma (PTCL) |
| 12.3 | Key PTCL Market Forecast Assumptions |
| 12.4 | Conjoint Analysis of Peripheral T-cell lymphoma (PTCL) |
| 12.5 | Total Market Size of Peripheral T-cell lymphoma (PTCL) in the 7MM |
| 12.6 | Total Market Size of Peripheral T-cell lymphoma (PTCL) by Therapies in the 7MM |
| 12.7 | The United States PTCL Market |
| 12.7.1 | Total Market Size of Peripheral T-cell lymphoma (PTCL) in the United States |
| 12.7.2 | Total Market Size of Peripheral T-cell lymphoma (PTCL) by Therapies in the United States |
| 12.8 | EU4 and the UK PTCL Market |
| 12.1 | Japan PTCL Market |
| 13 | Unmet Needs of Peripheral T-cell lymphoma (PTCL) |
| 14 | SWOT Analysis of Peripheral T-cell lymphoma (PTCL) |
| 15 | KOL Views of Peripheral T-cell lymphoma (PTCL) |
| 16 | Market Access and Reimbursement of Peripheral T-cell lymphoma (PTCL) |
| 16.1 | The United States |
| 16.2 | In EU4 and the UK |
| 16.3 | Japan |
| 16.4 | Summary and Comparison of Market Access and Pricing Policy Developments in 2025 |
| 16.5 | Market Access and Reimbursement of Peripheral T-cell lymphoma (PTCL) Therapies |
| 17. | Bibliography |
| 18 | Peripheral T-Cell Lymphoma Market Report Methodology |
Related Reports
Peripheral T-Cell Lymphoma Clinical Trial Analysis Pipeline
Peripheral T-Cell Lymphoma Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key Peripheral T-Cell Lymphoma companies, including Acrotech Biopharma LLC, HUYA Bioscience International, Seattle Genetics, Inc., Akeso Pharmaceuticals, Inc., Shandong New Time Pharmaceutical Co., LTD, Verastem, Inc., Genor Biopharma Co., Ltd., CerRx, Inc., Shanghai YingLi Pharmaceutical Co., Ltd., Dizal Pharmaceuticals, Kura Oncology, Inc., and others.
Non-Hodgkin’s Lymphoma Market
Non-Hodgkin’s Lymphoma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key non-Hodgkin’s lymphoma companies, including Soligenix, Innate Pharma, Nurix Therapeutics, AstraZeneca, Merck, Prescient Therapeutics, Kite Pharma, Seagen, Takeda, Novartis, Daiichi Sankyo, Genmab, AbbVie, Genentech (a Member of Roche), Bristol Myers Squibb, and others.
Cutaneous T-cell Lymphoma Market
Cutaneous T-cell Lymphoma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key CTCL companies, including Soligenix and Sterling Pharma Solutions, Prescient Therapeutics, Innate Pharma, Bristol-Myers Squibb, ONO Pharmaceutical, and others.
Follicular Lymphoma Market
Follicular Lymphoma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key follicular lymphoma Market companies, including Merck Sharp and Dohme, AstraZeneca, CRISPR Therapeutics, BeiGene, Nektar Therapeutics, NovalGen, Carna Biosciences, Allogene Therapeutics, Xynomic Pharmaceuticals, Bristol-Myers Squibb, Incyte Corporation, among others.
Diffuse Large B-cell Lymphoma Market
Diffuse Large B-cell Lymphoma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key DLBCL companies, including Roche (Genentech), Biogen, Nektar Therapeutics, Merck, Allogene Therapeutics, Miltenyi Biomedicine, AstraZeneca, BioVaxys, ImmunoVaccine Technologies, Cellectar Biosciences, Galapagos, Novartis, Lyell, ImmPACT Bio, Pfizer, Kartos Therapeutics, 2seventy bio, Regeneron Pharmaceuticals, BeiGene, Ranok Therapeutics, Constellation Pharmaceuticals, Genmab, IDP Discovery Pharma S.L., Immunitas Therapeutics, Monte Rosa Therapeutics, SymBio Pharmaceuticals, AVM Biotechnology, Autolus Therapeutics, Kymera Therapeutics, Otsuka Pharmaceutical, Caribou Biosciences, Adicet Bio, Gilead Sciences, Xynomic Pharmaceuticals, Amgen, among others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur info@delveinsight.com +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaEveningPost.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaEveningPost.com takes no editorial responsibility for the same.
