Global Biosimulation Market Poised for Phenomenal Expansion at a CAGR of ~17% by 2034 | DelveInsight

The biosimulation market is witnessing robust expansion, fueled by the increasing adoption of model-informed drug development (MIDD), the growing complexity of modern drug development, and the rising emphasis on personalized and precision medicine. MIDD strategies allow pharmaceutical companies to leverage advanced computational models to forecast drug behavior, refine dosing strategies, and minimize dependence on expensive clinical trials, ultimately speeding up development timelines.
New York, USA, April 27, 2026 (GLOBE NEWSWIRE) — Global Biosimulation Market Poised for Phenomenal Expansion at a CAGR of ~17% by 2034 | DelveInsight
The biosimulation market is witnessing robust expansion, fueled by the increasing adoption of model-informed drug development (MIDD), the growing complexity of modern drug development, and the rising emphasis on personalized and precision medicine. MIDD strategies allow pharmaceutical companies to leverage advanced computational models to forecast drug behavior, refine dosing strategies, and minimize dependence on expensive clinical trials, ultimately speeding up development timelines.
DelveInsight’s Biosimulation Market Insights report provides the current and forecast market analysis, individual leading biosimulation companies’ market shares, challenges, biosimulation market drivers, barriers, trends, and key biosimulation companies in the market.
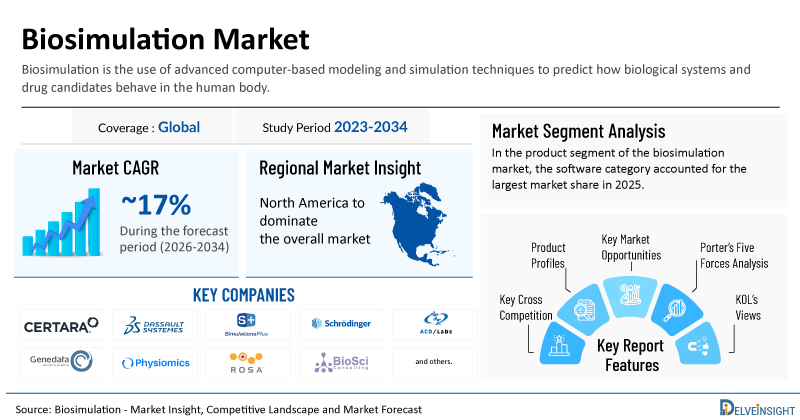
Biosimulation Market Summary
- 2025 Biosimulation Market Size: USD 4.7 Billion
- 2034 Projected Biosimulation Market Size: USD 20 Billion
- Biosimulation Market Growth Rate (2026-2034): ~17%
- Largest Biosimulation Market: North America
- Largest Product Segment: Software Category
- Key Companies in the Biosimulation Market: Certara, Inc., Dassault Systèmes (BIOVIA), Simulations Plus, Inc., Schrödinger, Inc., Advanced Chemistry Development (ACD/Labs), Genedata AG, Physiomics Plc, Rosa & Co. LLC, BioSimulation Consulting Inc., InSilico Biotechnology AG, Cadence, and Cellworks. life, and others
To read more about the latest highlights related to the biosimulation market, get a snapshot of the key highlights @ https://www.delveinsight.com/sample-request/biosimulation-market
Key Factors Contributing to the Rise in Growth of the Biosimulation Market
- Rising Need to Reduce Drug Development Time and Cost: Traditional drug development is expensive, time-consuming, and carries a high failure rate. Biosimulation helps companies predict drug behavior, optimize dosing, and identify potential risks early, reducing late-stage trial failures and accelerating approvals.
- Increasing Adoption of Precision Medicine: The growing focus on personalized therapies has increased demand for patient-specific modeling tools. Biosimulation enables researchers to simulate treatment responses across diverse patient populations, supporting tailored therapies and better outcomes.
- Advancements in AI, Machine Learning, and HPC: Integration of artificial intelligence, machine learning, and high-performance computing has significantly improved the speed, accuracy, and scalability of biosimulation platforms. These technologies enable faster analysis of complex biological systems and large datasets.
- Strong Growth in Pharmaceutical and Biotechnology R&D Spending: Increasing R&D investments by pharma and biotech companies are fueling the adoption of biosimulation solutions for biologics, gene therapies, oncology drugs, and rare disease treatments.
- Regulatory Support for Model-Informed Drug Development (MIDD): Regulatory agencies such as the FDA and EMA are increasingly accepting simulation-based evidence in submissions. This support encourages companies to integrate biosimulation throughout the drug development lifecycle.
- Expansion of Virtual Clinical Trials and In Silico Testing: The industry is shifting toward virtual trials and in silico testing to reduce dependence on costly human trials, improve trial design, and shorten development timelines.
- Growing Complexity of Modern Therapeutics: New modalities such as biologics, cell therapies, gene therapies, and immunotherapies require advanced modeling to understand pharmacokinetics, toxicity, and efficacy. Biosimulation provides critical support in these complex areas.
- Increasing Adoption of Cloud-Based Platforms: Cloud deployment models offer scalability, lower infrastructure costs, and easier collaboration across global research teams, making biosimulation more accessible to companies of all sizes.
Get a sneak peek at the biosimulation market dynamics @ Biosimulation Market Trends
Regional Biosimulation Market Insights
North America
- North America held the largest share of the biosimulation market in 2025, contributing 44.24% of the global market.
- This leadership is driven by the region’s well-established pharmaceutical and biotechnology sector, sophisticated healthcare infrastructure, substantial investments in research and development, and broad adoption of computational technologies for drug discovery and development.
- The region is home to several prominent biosimulation and life sciences technology companies, including Certara, Simulations Plus, and Schrödinger, Inc., all of which continue to advance innovative modeling solutions for pharmaceutical research.
- Moreover, strong regulatory backing from the U.S. Food and Drug Administration for model-informed drug development (MIDD) has further encouraged pharmaceutical companies to adopt biosimulation tools for clinical trial planning, dose optimization, and safety assessments.
Europe
- In Europe, the biosimulation market is experiencing strong growth due to favorable regulatory policies, rising adoption of model-informed drug development, and robust partnerships between pharmaceutical companies and technology providers.
- Regulatory agencies such as the European Medicines Agency are significantly contributing to this expansion by encouraging the integration of biosimulation into drug development and approval pathways.
- For example, in May 2024, the EMA endorsed the use of biosimulation data for regulatory decision-making in marketing authorization applications, representing an important milestone in the broader acceptance of simulation-driven approaches.
Asia-Pacific
- The Asia Pacific region is becoming a major contributor to growth in the biosimulation market, driven by the rapid expansion of pharmaceutical and biotechnology research activities, supportive government initiatives, and increasing use of advanced computational modeling technologies in countries like China, India, and Japan.
- The region is further strengthened by growing investments in precision medicine, AI-enabled drug discovery, and affordable in silico modeling solutions, which help accelerate and improve the accuracy of drug development.
- Governments across the region are also promoting this transition. For example, India’s National Biotechnology Development Strategy (2021–2025) has committed significant funding to enhance digital and computational infrastructure in drug research, boosting the adoption of biosimulation tools nationwide.
To know more about why North America is leading the market growth in the biosimulation market, get a snapshot of the Biosimulation Market Share
Recent Developmental Activities in the Biosimulation Market
- In March 2026, Certara, Inc. announced the launch of Simcyp® Simulator Version 25, the latest update to its physiologically-based pharmacokinetic (PBPK) modeling platform used to simulate drug behavior in virtual populations, thereby streamlining regulatory submissions and drug development.
- In October 2025, Certara launched Certara IQ™, an AI-powered Quantitative Systems Pharmacology (QSP) biosimulation platform, which integrates artificial intelligence with systems biology models to improve drug-disease modeling, optimize dosing strategies, and enhance pharmaceutical R&D productivity.
- In August 2025, Certara’s Simcyp® Simulator became one of the first platforms to receive formal EMA qualification for physiologically based pharmacokinetic (PBPK) modeling, enabling its use in regulatory submissions across Europe.
What is Biosimulation?
Biosimulation is the use of advanced computer-based modeling and simulation techniques to predict how biological systems and drug candidates behave in the human body. It combines data from biology, pharmacology, physiology, and clinical studies to create virtual models that can estimate a drug’s absorption, distribution, metabolism, efficacy, safety, and potential outcomes across different patient populations. Widely used in pharmaceutical and biotechnology industries, biosimulation supports decision-making throughout drug development by optimizing dose selection, reducing trial risks, improving study design, and accelerating timelines while lowering costs. Common applications include physiologically based pharmacokinetic (PBPK) modeling, population pharmacokinetic/pharmacodynamic (PK/PD) analysis, and disease progression modeling, making biosimulation a critical tool in modern precision medicine and regulatory science.
| Biosimulation Market Report Metrics | Details |
| Coverage | Global |
| Study Period | 2023–2034 |
| Biosimulation Market CAGR | ~17% |
| Biosimulation Market Size by 2034 | USD 20 Billion |
| Key Biosimulation Companies | Certara, Inc., Dassault Systèmes (BIOVIA), Simulations Plus, Inc., Schrödinger, Inc., Advanced Chemistry Development (ACD/Labs), Genedata AG, Physiomics Plc, Rosa & Co. LLC, BioSimulation Consulting Inc., InSilico Biotechnology AG, Cadence, and Cellworks. life, and others |
Biosimulation Market Assessment
- Biosimulation Market Segmentation
- Biosimulation Market Segmentation By Component: Software and Services
- Biosimulation Market Segmentation By Deployment Mode: Cloud-based and On-premises
- Biosimulation Market Segmentation By Application: Drug Discovery and Development, Disease Modelling, Pharmacokinetics/Pharmacodynamics (PK/PD) Modeling, and Others
- Biosimulation Market Segmentation By Delivery Model: Subscription Models, Ownership Models, and Service-Based Models
- Biosimulation Market Segmentation By End User: Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Others
- Biosimulation Market Segmentation By Geography: North America, Europe, Asia-Pacific, and Rest of World
- Porter’s Five Forces Analysis, Product Profiles, Case Studies, KOL’s Views, Analyst’s View
Which MedTech key players in the biosimulation market are set to emerge as the trendsetter explore @ Biosimulation Market Analysis
Table of Contents
| 1 | Biosimulation Market Report Introduction |
| 2 | Biosimulation Market Executive Summary |
| 3 | Biosimulation Market Key Factors Analysis |
| 4 | Impact Analysis |
| 5 | Regulatory Analysis |
| 6 | Biosimulation Market Porter’s Five Forces Analysis |
| 7 | Biosimulation Market Assessment |
| 8 | Competitive Landscape |
| 9 | Startup Funding & Investment Trends |
| 10 | Biosimulation Market Company and Product Profiles |
| 11 | KOL Views |
| 12 | Project Approach |
| 13 | About DelveInsight |
| 14 | Disclaimer & Contact Us |
Interested in knowing the biosimulation market share by 2034? Click to get a snapshot of the Biosimulation Market Size
Related Reports
Contract Development Manufacturing Organization Market
Contract Development Manufacturing Organization Market Insights, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key CDMO companies, including Patheon (Thermo Fisher Scientific), Catalent,Inc, Laboratoire Elaiapharm, The Lubrizol Corporation, Cambrex Corporation, Recipharm (EQT), Merck & Co., Inc, AGC Biologics, Nerpharma S.r.l, Pfizer CenterOne (Pfizer Inc), EMERGENT, Onyx Scientific Limited, Lonza, Siegfried Holding AG, CordenPharma International, FUJIFILM Diosynth Biotechnologies, Samsung Biologics, Delpharm, Center for Breakthrough Medicines, and WuXi Biologics, and others.
Biopharmaceuticals Contract Manufacturing Market
Biopharmaceuticals Contract Manufacturing Market Insights, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key biopharmaceuticals contract manufacturing companies, including Boehringer Ingelheim International GmbH, Lonza, Rentschler Biopharma SE, BINEX Co., Ltd., INCOG BioPharma Service, FUJIFILM Diosynth Biotechnologies U.S.A. Inc., Novartis AG, ProBioGen AG, TOYOBO CO., LTD., Samsung Biologics, Thermo Fisher Scientific, Inc., AbbVie, Inc., WuXi Biologics, AGC Biologics, ADMA Biologics, Inc., Baxter BioPharma Solutions (Baxter), iBio., Catalent, Inc., Cambrex Corporation, Pfizer Inc., and others.
Biologics Drug Substance Fill and Finish Manufacturing Market
Biologics Drug Substance Fill and Finish Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key biologics drug substance fill and finish manufacturing companies, including JOINN Biologics, Pharmaron, Symbiosis Pharmaceutical Services, AGC Biologics, INCOG BioPharma Services, CPC, Eurofins Scientific, and others.
Biologics Drug Substance Manufacturing Market
Biologics Drug Substance Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key biologics drug substance manufacturing companies, including Lonza, Pfizer Inc., Catalent, Inc., Boehringer Ingelheim International GmbH, Samsung Biologics, WuXi AppTec, Thermo Fisher Scientific Inc., Asahi Kasei Corporation, Recipharm AB, Evotec, Novo Nordisk A/S, Siegfried Holding AG, Enzene Biosciences Ltd., Forge Biologics, BeiGene LTD., FUJIFILM Holdings Corporation, TOYOBO CO., LTD., AJINOMOTO CO., INC., Biocon, Eurofins Scientific, and others.
Biologics Contract Manufacturing Market
Biologics Contract Manufacturing Market Insights, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key biologics contract manufacturing companies, including Boehringer Ingelheim, Catalent, Lonza, WuXi Biologics, Samsung Biologics, FUJIFILM Diosynth Biotechnologies, AbbVie, AGC Biologics, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur info@delveinsight.com +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaEveningPost.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaEveningPost.com takes no editorial responsibility for the same.
